The “hydrogen economy” is currently hindered by the low rate and efficiency of hydrogen production from water and energy generation by hydrogen oxidation into water. The former process is controlled by the Hydrogen Evolution Reaction (HER), the cathodic reaction during the electrochemical process of dissociating the water molecule into hydrogen and oxygen. The latter is controlled by the kinetics of the cathode Oxygen Reduction Reaction (ORR). The sluggish kinetics of both the HER and ORR can only be improved by increasing the reaction temperature or using Pt catalysts. Therefore, there is a strong need for cheaper and more durable catalysts to replace Pt.
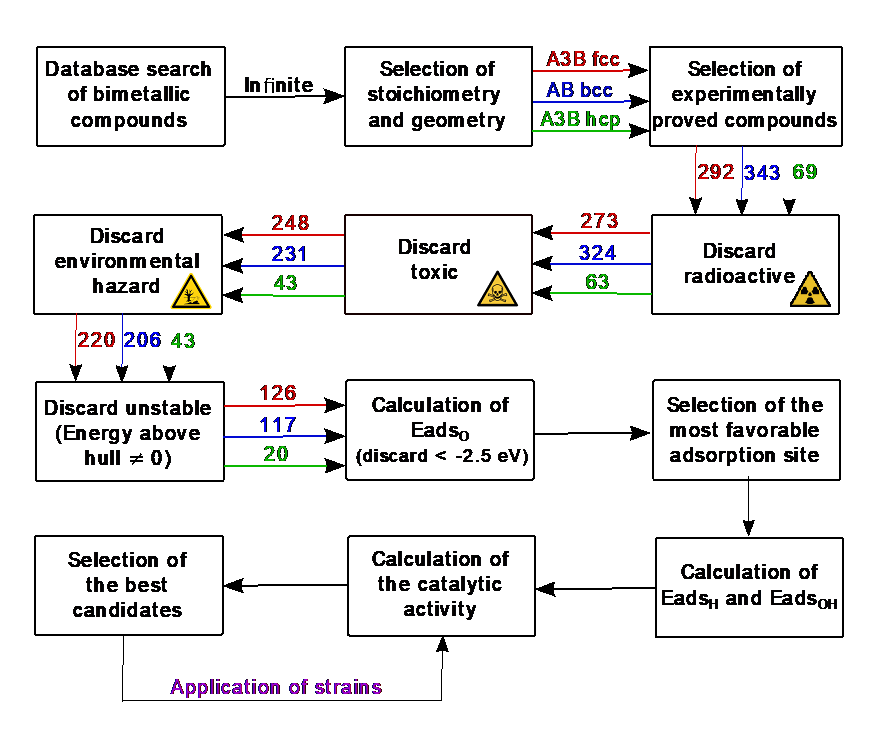
IMDEA Materials has developed a new high-throughput strategy to search for new intermetallic compounds that can be used to replace Pt for both reactions. The method is based on first-principles calculations and elastic strain engineering [1,2]. The basic idea is to develop fast machine learning algorithms that accurately predict the adsorption energies for different intermediate species in the catalytic reaction from descriptors that can be easily obtained for different catalysts. Descriotors include geometrical features (surface geometry, binding site, neighboring atoms, etc.), element properties (electronegativity, valence electrons, electron affinity), as well as other properties obtained from first principles calculations (surface energy, d-band center, etc.). All these descriptors are available in large repositories, such as the Materials Project, OQMD, AFLOW or the Catalysis Hub. The optimum descriptors and the most suitable machine learning algorithms are developed by calculating the catalytic activities for both HER and ORR through density functional theory simulations for a large enough number of catalysts used to train the model, including the effect of elastic strains. The trained model is used afterwards to explore a much larger space of catalysts to find new options.
The optimum intermetallic compounds and elastic strains for either HER or ORR are then synthetized by means of magnetron sputtering on suitable substrates, and elastic strains are introduced by choosing the thermo-mechanical properties of the intermetallic compound and substrate and the deposition temperature or by the application of mechanical deformations. Catalytic properties are finally assessed through standard electrochemical techniques such as the cyclic and linear sweep voltammetry.
[1] C. Martínez-Alonso, J. M. Guevara-Vela, J. LLorca. The effect of elastic strains on the adsorption energy of H, O and OH in transition metals. Physical Chemistry Chemical Physics, 23, 21295-21306, 2021.
