Published by: Dr. David Tilve Martínez. PhD in Physical Chemistry of Condensed Matter.
The original version of this article can be found in Spanish in Fundación Muy Interesante.
Can you imagine a T-shirt stronger than steel, more conductive than copper, and as light as silk? This is no longer science fiction. Research into new materials is exploring fibres based on physicochemical phenomena that once inspired Kevlar.
Today, Kevlar remains one of the strongest fibres on the market: it is used in bulletproof vests, helmets, and seat belts, and its industrial production reaches thousands of tonnes per year. Its manufacture involves a peculiar state of matter that is neither solid nor liquid, but something in between: a liquid crystal. Today, that same principle is being applied to nanomaterials capable of revolutionising technical textiles and flexible electronics.
A glass of water resting on a table appears motionless. Yet at the molecular scale, its particles constantly move and collide in every direction. This perpetual “dance” is known as Brownian motion and is completely random. Now imagine that, instead of small spherical molecules, we introduce tiny rigid needle-shaped rods into the water. If they are small enough, they will also move freely: rotating and translating without any preferred direction.
But if we add many more of them, space begins to run out, and each rod collides with the others. Eventually, the only way they can continue moving without blocking one another is to align spontaneously. They do not become a rigid solid, but they do begin to point, on average, in the same direction. This intermediate state, order in orientation combined with freedom of movement, is a liquid crystal.
A simple analogy is that of a lake filled with logs. When there are only a few, they float and rotate without coordination. But as the lake fills up, the logs eventually group together into aligned bundles in order to fit. The order does not arise from their composition, but purely from geometric necessity. And this partial order can be exploited technologically.

Can nanomaterials behave as rigid rods?
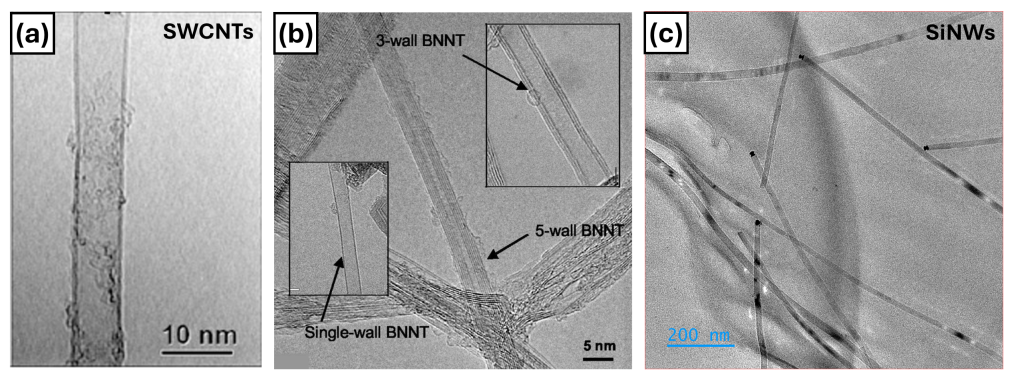
In many cases, the answer is yes. In particular, carbon nanotubes (CNTs), boron nitride nanotubes (BNNTs), and silicon nanowires (SiNWs) share one important feature: their elongated, one-dimensional (1D) shape. They are thousands of times thinner than a human hair and between one hundred and one million times longer than they are wide.
This geometry makes them ideal candidates to behave like “rods”. Carbon nanotubes, for example, can be understood as rolled-up carbon sheets with diameters of only a few nanometres (a millimetre divided one million times). Individually, they exhibit extraordinary mechanical and electrical properties: they are incredibly strong and excellent conductors of electricity and heat.
The problem arises when trying to use trillions of them to manufacture a macroscopic material. If they are mixed without control, they become entangled like a chaotic ball of yarn. Their individual properties are not automatically transferred to the bulk material. This is where liquid crystals come back into the picture. At sufficiently high concentrations in a suitable solvent, these elongated nanomaterials can self-organise into an ordered phase. This is the crucial step between the nanoscale and the visible world.
Why all this complexity just to make fibres?
At the nanoscale, some materials behave in almost extraordinary ways. A single carbon nanotube can be stronger than steel for the same weight and conduct electricity better than many metals. Boron nitride nanotubes stand out for their thermal stability and chemical resistance. Silicon nanowires possess one of the highest refractive indices among known materials.
In the laboratory, these properties are measured in individual nanomaterials that are nearly perfect. In practice, however, these nanomaterials are usually obtained as powders: disordered aggregates in which nanotubes or nanowires are entangled and randomly oriented. In this disordered state, many of their exceptional properties are diluted because of poor compaction. It is like trying to build a structure using bricks with no defined orientation: the force cannot be transmitted properly.
The key is not only to have extraordinary “bricks”, but also to know how to organise them. If those same nanotubes or nanowires are used as building blocks and compacted in an aligned way, the result changes dramatically.
When they all point in the same direction, mechanical load is distributed uniformly, electricity flows with fewer obstacles, and heat is transmitted more efficiently along the fibre axis. The difference between a disordered powder and an aligned fibre is not merely structural: it is functional. This is where the liquid crystal phase becomes essential. Instead of attempting to order the nanostructures once they are already solid and disordered, researchers exploit their ability to self-organise while still in a fluid state.
At suitable concentrations, they become pre-ordered, allowing their orientation to be fixed during the spinning process. When the fluid is injected through a nozzle into a coagulation bath and solidifies, trillions of nanostructures remain aligned along the fibre axis. In this way, their individual properties cease to be laboratory curiosities and become expressed in a macroscopic material. In other words, the challenge is not to manufacture extraordinary nanomaterials, that is already possible, but to translate their properties from the nanoscale to the visible scale. And the liquid crystal is the translator.

Applications from everyday life to space
The turning point came at the beginning of this century. In 2000, French researcher Philippe Poulin demonstrated that carbon nanotubes could behave as rigid rods capable of forming liquid crystal phases and fibres. That idea enabled the fabrication of the first macroscopic fibre based on aligned nanotubes. With this work, an entire field was opened: the goal was no longer simply to synthesise extraordinary nanomaterials, but to organise them collectively so that their properties could emerge at large scale.
For years, progress remained mainly academic. But two decades later, companies are beginning to emerge that aim to bring this macromolecular architecture to market. One example is DexMat, which develops conductive fibres based on aligned carbon nanotubes. Their materials combine extreme lightness with high electrical conductivity and mechanical strength, making them candidates for ultralight cables in electric motors and electromagnetic shielding in aerospace structures.
As mentioned in the previous section, if we change the building block, we change the final function of the fibre. With carbon nanotubes (CNTs), the focus is on mechanical strength and electrical conductivity. But the same approach can be applied to other nanomaterials. Boron nitride nanotubes (BNNTs), for example, are electrically insulating and excellent thermal conductors. In addition, they show remarkable resistance to gamma radiation, making them attractive for space environments where there is no atmosphere to protect us.
Silicon nanowires (SiNWs) could enable fibres capable of acting as distributed electronic devices within a textile: sensors, efficient light transmission systems, or even integrated energy storage systems. Imagine flexible batteries woven directly into technical clothing.

An order born from disorder
The physicochemistry that makes these fibres possible does not change. The only thing that changes is the “brick” used in the liquid crystal phase before spinning. If in the twentieth century this development enabled the production of fibres such as Kevlar, the twenty-first century may be defined by the engineering of functional fibres: not only strong, but also electrically conductive, thermally conductive, magnetic, or energetically active. The challenge remains the same as it was twenty years ago: achieving perfect alignment, minimising defects, and scaling up production without losing the properties that make these materials special at the nanoscale.
Paradoxically, the future of textiles may depend on a state of matter that emerges from chaos. Molecules and nanostructures moving freely, which upon reaching a certain concentration discover that alignment is the best way to coexist. This delicate balance between freedom and order, neither solid nor liquid, will give rise to the smart materials of the twenty-first century.
From the Multifunctional Nanocomposites Group at IMDEA Materials Institute, we believe that the next textile revolution will not come from exotic new polymers, but from learning how to better organise what we already know how to manufacture. Because sometimes the secret is not to invent something new, but to arrange what already exists.
References
- [1] Paineau et al. «A liquid-crystalline hexagonal columnar phase in highly-dilute suspensions of imogolite nanotubes.» Nature Communications 7.1 (2016): 10271.
- [2] Smith et al. «Very long single-and few-walled boron nitride nanotubes via the pressurized vapor/condenser method» Nanotechnology 20.50 (2009): 505604.
- [3] Vigolo et al. «Macroscopic fibers and ribbons of oriented carbon nanotubes.» Science 290.5495 (2000): 1331-1334.
- [4] Miaudet et al. Hot-drawing of single and multiwall carbon nanotube fibers for high toughness and alignment. Nano letters 5.11 (2005): 2212-2215.
- [5] https://dexmat.com/
- [6] https://www.bnnt.com/
The original version of this article can be found in Spanish in Fundacion Muy Interesante.
