Although secondary Li-ion batteries are widely used for electrochemical energy storage, low energy (100-300 Wh kg-1) and power density (250-400 W kg-1) are limiting their applications in several areas including long-range electric vehicles.
This is mainly due to the use of graphite anodes with low specific capacity (<372 mAh g-1) and sluggish Li-ion diffusion kinetics. Lithium-ion intercalation of graphite anodes at lower potentials (<0.2 V vs Li+/Li) can also results in dendrite growth and associated short circuit.
These drawbacks of graphite anodes triggered extensive research focused on the development of alternative high-performance and safer anode materials. Despite of the high specific capacities, conversion and alloying type electrodes (SnO2, Fe2O3, Co3O4, NiO, MnO2, WO3, Si, Ge, Sn etc.) experienced severe capacity fading on extended cycling.
Diffusion independent pseudocapacitive ion storage is one of the recently investigated mechanisms for achieving ultrafast Li-ion storage. However, pseudocapacitive type Li-ion storage is limited in the case of conversion, alloying and insertion type transition metal oxide anodes.
While pseudocapacitance can be induced by nanostructuring (extrinsic pseudocapacitance), this method remains elusive in the case of metal oxide anodes.
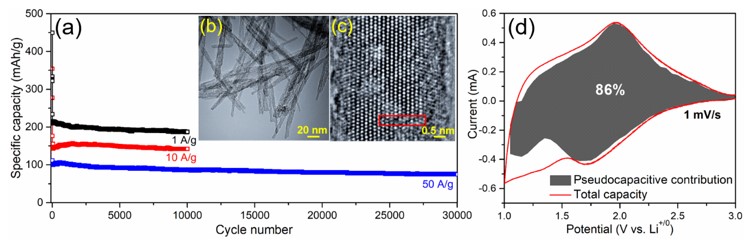
In order to mitigate these issues, IMDEA Materials has developed defect-engineered (0D, 1D and 2D defects) metal-oxide anodes for high energy/ power density Li-ion batteries. For instance, oxygen deficient rutile-TiO2 nanowires have been demonstrated as a pseudocapacitive anode for ultrafast charging lithium-ion batteries (Fig. 1).
Li-ion batteries capable of superfast charging (up to 7s)and ultralong life (up to 30,000 cycles) can be fabricated based on the newly defect nanoengineered anodes. This strategy can also be extended for numerous transition metal oxide electrodes used in Li and Na-ion batteries.