Diffusion independent pseudocapacitive ion storage is one of the recently investigated mechanisms for achieving ultrafast Li and Na-ion storage. It usually involves surface/ near surface charge-transfer reactions. Nevertheless, intrinsic pseudocapacitance of transition metal oxide anodes is not sufficient to deliver high energy and power densities.
Although pseudocapacitance can be induced by nanostructuring and defect/ interface engineering, this method remains elusive in the case of metal oxide anodes.
Nanoscale engineering is a potential solution to circumvent the challenges associated with conventional lithium-ion battery electrodes because of their diverse surface properties, tunable structures and improved conductivity.
IMDEA Materials has developed defect-engineered (0D, 1D and 2D defects) metal-oxide anodes for high energy/ power density Li/ Na-ion batteries.
Li and Na-ion storage mechanism in most of the pseudocapacitive electrodes reported have not been systematically investigated.
Improved performances are often assigned to surface storage, and the exact the mechanism is not clear. Advanced characterization techniques are therefore required to explain the crucial reasons for the defect engineered pseudocapacitive electrodes.
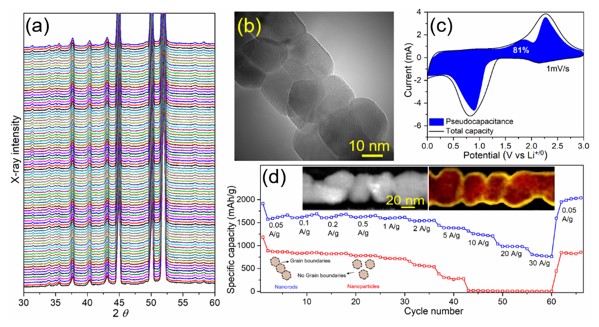
For instance, IMDEA Materials has investigated pseudocapacitive Li-ion storage mechanisms (see Fig. 1) enabled by nanograin-boundaries of polycrystalline Co3O4 nanorods through in-situ X-ray diffraction and Electron Energy Loss Spectroscopy (EELS) analysis.
These in-situ measurements can be also employed to investigate the Li/ and Na-ion storage mechanism of a variety of electrode materials.